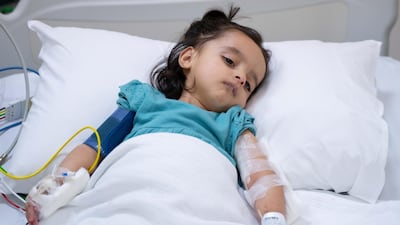
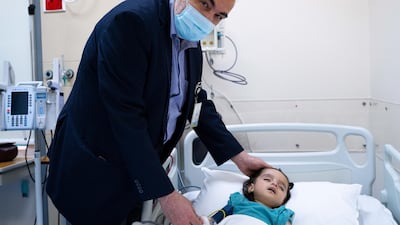
A specialised team of paediatric neurologists at Sheikh Khalifa Medical City (SKMC) in Abu Dhabi have successfully treated a 17-month-old girl with spinal muscular atrophy (SMA).
The clinicians used a range of advanced treatments, including gene therapy medicine brought in from abroad.
Doctors diagnosed Afra's condition after her mother noticed she was making abnormal movements when she was three months old.
Dr Omar Ismail, head of paediatric neurology at SKMC, said this helped her to recover.
“The key to successfully overcoming SMA is early diagnosis and implementation of a vigorous treatment strategy," he said.
"This is to keep the nerve's cells, or neurons, as intact as possible and prevent further damage.
"In Afra’s case, we quickly jumped into action with an inclusive treatment plan that prevented further deterioration of her muscles while we waited for the required genetic treatment to arrive from abroad.
"This particularly helped with Afra’s breathing muscles and eliminated the need for an artificial respiratory device.”
SMA is a group of inherited diseases that damage nerve cells in the spinal cord, called motor neurons.
This damage is progressive and affects actions like swallowing, breathing, sitting, and walking.
SMA is passed on to children by parents through abnormal genes, with the most common form of SMA caused by an unusual or missing survival motor neuron 1 gene (SMN1 gene).
If left untreated, the disease can lead to death and the need for permanent ventilation by the age of 2 in more than 90 per cent of cases.
Although it is impossible to cure the disorder, revolutionary new treatments have been introduced in recent years to help manage symptoms, reduce discomfort, and maintain quality of life.
The genetic treatment provided to Afra cures the root cause of the disease with a one-time infusion that replaces the defective or missing SMN1 gene with a working copy.
Thereafter, the muscle will continue to work well with improving movement and functions.
The medicine was only recently approved by the US Food and Drug Administration for use in children less than 2 years of age.
Dr Mariam Buti Al Mazrouei, chief executive director at the hospital, which is part of the Abu Dhabi Health Services Company (Seha) network, said they managed to import the medicine quickly.
"Thanks to the UAE’s robust and ever-expanding healthcare landscape we have fast, simple and streamlined access to integral tools and resources from abroad," she said.
"Afra’s journey towards recovery is a significant achievement, not just for the Seha network but for the wider healthcare ecosystem in the country, as we provide hope and create impact for families with children diagnosed with SMA.”
Afra's mother said she appreciated the fast actions of the medical practitioners who saved her daughter's life.
“I am tremendously grateful to the team at SKMC and the wider Seha network for their diligence in treating my daughter,” she said.