Dubai's Medcare Royal Speciality Hospital has become the first outside the US to administer Itvisma, a new drug that treats spinal muscular atrophy, to an adult patient.
The patient, a 22-year-old Egyptian man, travelled to the UAE to receive the one-time gene-therapy, which is priced at $2 million a dose.
About four months ago, three-year-old Hulus from Turkey received the drug at Medcare Women and Children Hospital in Dubai in the first use of the treatment outside the US, shortly after UAE regulators approved the medicine.
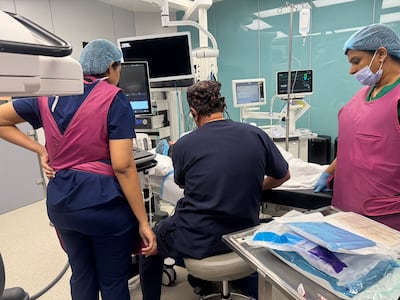
The Egyptian man, who has spent most of his life in a wheelchair, had Itvisma injected into his cerebrospinal fluid, which circulates through the spine and around the brain.
Dr Vivek Mundada, clinical lead of the gene therapy unit at Medcare Hospitals in the UAE, told The National he was “very, very pleased” to administer the treatment.
“Because it’s a gene therapy, we are addressing the root cause,” he said. “The natural course of this disease is dreadful if you don’t treat it things will go downhill.
“Even if I can achieve the stability and the strength – the strength in the arms, the torso, the muscles in the neck – if we can maintain that strength, I would be very happy in this case.”
The patient is only the second adult in the world to receive Itvisma, which is produced by Novartis.
Complex case
Administering the treatment was complex because SMA had caused the patient to develop scoliosis, or curvature of the spine, which doctors had previously attempted to treat through surgery.
X-rays were used to guide surgeons in administering the injection. Once the needle was in place, it took about five minutes to inject the 3ml dose, Dr Mundada said.
“Thankfully it was painless, there were not any complications afterwards,” he added.

Itvisma provides a replacement for the patient’s defective survival motor neuron 1 gene. Patients with the defective gene do not produce survival motor neuron protein normally, which impairs the action of motor neurons, nerve cells that transmit the brain’s instructions to the muscles.
This causes weakness in the limbs, often meaning that patients find walking impossible or difficult, and may have problems swallowing or breathing.
Being a neurodegenerative condition, untreated SMA often causes worsening complications as a patient ages.
Diagnosed at early age
The patient had SMA diagnosed at 18 months old, and had been receiving injections of a different drug, Spinraza, every four months. These will no longer be necessary.
A finance graduate, the patient is scheduled to begin a new job in the next few months, Dr Mundada said.
In a statement released by Medcare, the patient said the experience of receiving the injection had been “very positive”.
“I was constantly well informed, with the whole team taking very good care of me and being extremely responsive. This eased the process and made it smooth,” he said. “The injection itself was also easy, with no pain felt around the injection area. I am just glad that I do not have to take multiple injections like I used to before.”
When Itvisma is used to treat children, improvements in function may be achieved after the drug is administered, because their bodies are still growing.
Another gene therapy treatment, Zolgensma, was launched before Itvisma, but is suitable only for infants and children aged up to two years old.
SMA exists in various forms, the most serious of which, type 1, develops in the first six months of a patient's life and is usually fatal without treatment.
Type 2 may prevent a patient from being able to walk, while types 3 and 4 produce effects that are less severe but still significant.
The price of the Itvisma treatment, including aftercare and other costs, approached $2 million, Dr Mundada said. In this case, the therapy was funded by the patient’s father, a businessman.
Once branded the world's most expensive drug, Itvisma has now been surpassed in cost by other gene therapies, Dr Mundada said.
The cost of the treatment is expected to lower in the future.
Other cases
More patients are expected to be treated with Itvisma at Medcare Royal Specialty Hospital. This week, Dr Mundada has seen a 29-year-old man with a milder form of SMA, who could also receive the therapy. Two more patients are scheduled to be treated in May.
Dr Mundada will travel to Asia and Australia in the coming months to pass on his expertise as other hospitals look to begin administering the treatment.
“The experts in these countries want to understand how to implement this therapy,” he said. “Only the US and UAE are giving this treatment, and many people are wanting to learn.”
In a statement, Medcare said it had now treated more than 190 SMA patients, as well as 20 patients with Duchenne muscular dystrophy, which also causes muscle weakness and mobility problems. The company said Dubai had become “a genuine global destination” for gene therapy and related treatments.
SMA tends to be more common in the Gulf compared to the global average because of the tradition of consanguineous marriage, which makes it more likely that two people who carry the faulty gene will have children together. Measures such as premarital screening can reduce the condition’s prevalence.



